At $51.6 billion in revenue, global pharmaceutical powerhouse Pfizer is an undisputed industry leader. Operating in 175 markets and fielding 56 manufacturing sites, Pfizer provides consumers with a wide range of medicines and other therapies "from Advil to Zithromax,” explained Jose Medina, Pfizer senior analyst and intelligence manager, describing at RSTechED 2014 his company's 20-location rollout of FactoryTalk VantagePoint software from Rockwell Automation. Characterizing the company's product portfolio, Medina said that last year 10 brands were responsible for greater than $1 billion in sales.
A Challenge as Large as Pfizer Itself
A company the size of Pfizer has enormous challenges, said Medina, due to the complexity of manufacturing a broad range of pharmaceuticals that includes both patent-protected ones such as Viagra and biologics, as well as over-the-counter medications, which have an entirely different economic/production model dictating their profitability.
Regardless, since the introduction of the U.S. Food and Drug Administration's "Pharmaceutical GMPs for the 21st Century – A Risk-Based Approach” 11 years ago, the pharmaceutical industry has been faced with a regulatory regime based on Good Manufacturing Practices (GMP). In his article "Coming of Age” (Pharmaceutical Manufacturing, April 2013) author Angelo DePalma, noted "GMPs for the 21st Century has profoundly influenced how drug companies manage risk, particularly, but not exclusively, with regard to manufacturing. The risk-based approach becomes particularly challenging given the backdrop of merger and acquisition activity and a heavier reliance on outsourcing for manufacturing and R&D. For manufacturing, this means negotiating the moving targets of productivity and supply chain demands.”
It's within this context that Pfizer has steadily restructured and refined its manufacturing assets to support and implement GMPs, integrating them into to Pfizer Global Supply (PGS), the internal organization tasked with supplying the company with the products it develops and markets around the world. According to Pfizer Global Supply's John Kelly, vice president, sourcing & network strategy and transitioning operations, "Pfizer Global Supply [is] a critical element of Pfizer's success … We've always been a very vision-focused organization....Our purpose is to supply quality products for a healthier world.”
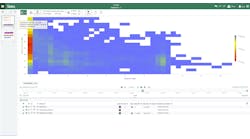
"What we wanted was a scalable, dynamic and value-adding information portal across the range of Pfizer core and manufacturing systems." Pfizer's Jose Medina on the company's successful deployment of FactoryTalk VantagePoint.
Fulfilling PGS's mission and vision, as Kelly succinctly described, is entirely dependent on managing its production assets as efficiently as possible, a task that is even more dependent on the operational truth derived from the enterprise manufacturing intelligence (EMI) it derives from the vast amounts of data streaming from its control devices and production and processing equipment and the subsequent reports Medina's department provides to PGS executive management. But, according to Medina, until recently, its reporting processes where clunky, slow and riddled with inefficiencies that were preventing PGS from obtaining in a timely fashion the critical KPIs and associated production metrics and statistics it needs to manage production assets effectively and profitably.Uphill Battle
Like Sisyphus, rolling the boulder up the mountain, only to watch it roll back down, then push it back again ad infinitum, Medina and his department's two other staff were responsible for the hundreds of reports PGS managers were demanding at every turn of each of the 56 location's business cycles. Medina explained that although the company implemented a core application that is the same at each location, each location's application is naturally adapted and localized to each site, which, in turn, thwarted the department's ability to provide the operational transparency to a central authority.
Maintaining commonality to create a unified reporting structure became an incredible challenge, said Medina. What came next was a litany of administrative issues associated with the logistics of connecting to each site to generate reports, meeting managers' informational needs locally, then translating those globally, etc., all in an effort to maintain report continuity across all of Pfizer's production assets. "So we have to keep those reports up to date among the sites, and if one site has a change, we have to ensure that we do the same change on every site. So we have to connect to each site globally and redeploy the report … and redeploy the report … and redeploy the report … and at the end of day, redeploying for each site takes us three or four days – perhaps a week to redeploy the entire suite of reports.”
Reporting issues didn't stop there either. Medina said it was virtually impossible for executive managers to obtain a common centralized view of all the production assets in one take (and in a timely fashion) because it still meant connecting to each site to obtain individual site reports. Even though Medina's team maintained report commonality, a common aggregated view or line-to-line comparisons were not available without more time-consuming manual intervention.
A Better Way
It became clearly evident that because of the company's incumbent reporting system, not only were Pfizer's data analysts being effectively stymied from efficiently delivering actionable information to the organization, no matter how much executive managers insisted, they also were not getting the information they needed for critical decision support up the chain of command. A true, unifying solution was needed. Fortunately FactoryTalk VantagePoint EMI from Rockwell Automation was ready to step in, creating the right solution at the right time for PGS.
"What we wanted to provide [PGS],” said Medina, "was a scalable, dynamic and value-adding information portal across the range of Pfizer core and manufacturing systems.” Adding that for business goals, "We needed to improve access to information, we needed to provide better understanding of the information, we needed the ability for users to create their own report scenarios, and we needed the ability for users to run trends for line and historic data.”
In terms of technical goals, said Medina, his department needed centralization and rapid deployment. "Previously it might have taken us one or two days to deploy a report. Of course, that meant we would have to ensure that report was copied over to all the sites. We wanted to be able to deploy reports to all the sites in one shot.” Ultimately, said Medina, his department created a platform that delivers standardized reports globally, removes the administrative work associated with variances and customization demanded by users, and supplies critical metrics and other knowledge when and where it is needed, on demand, ready to support data-confirmed regulatory compliance, product quality and the operational excellence mandatory for competitive success in the 21st century.