Operators, Engineers Collaborate to Boost Efficiency at Cellulose Fiber Plant
Figure 1: Adding model predictive control (MPC) to two large chlorine dioxide solution storage tanks essentially eliminated the abrupt production rate changes operators were making in an effort to manage the tank levels, allowing the methanol feed rate controller to stabilize the chlorine dioxide generator.
The computerized process control boom that occurred in the cellulose fiber (wood pulp) processing industry in the 1970s and 1980s was focused on increased plant productivity. This was achieved through increased production with a simultaneous workforce reduction. It's not uncommon to find pulp mills with production rates that far surpass nameplate capacities and only a fraction of the original number of employees. In most cases, increased plant throughput was unanimously cheered, whereas the workforce reductions generated bad feelings and fostered resistance to further automation projects. Unfortunately, this stigma persists to this day, even though job elimination through automation has become rare in pulp manufacturing. Automation projects now focus almost exclusively on efficiency, quality and safety. Process automation has become the good guy as it helps our fellow employees get home safe, save money for the company, and reduce our environmental footprint.This article presents both a human and technical perspective on a recent process control project at a Weyerhaeuser Co. pulp mill in Oglethorpe, Georgia. It was a project with excellent economic payback and numerous "soft” benefits for employees responsible for operating that unit. This was a complex project that demanded that we develop it with the operators, rather than just for them.
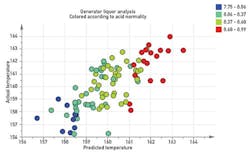
Figure 2. A regression model relating estimated concentration to generator temperature provides a continuous feedback signal the estimator can use to adjust concentration and efficiency estimates in response to unexpected temperature fluctuations.
Weyerhauser's Fling River Mill in Oglethorpe, Georgia, produces more than 350,000 tons per year of fluff pulp and specialty cellulose fiber products for a diverse group of customers. The cellulose fiber is recovered from wood chips in a large continuous digester, which is essentially a heated and pressurized plug flow reactor that dissolves lignin in caustic cooking liquor. Dissolved chemicals and lignin are separated from the fiber and recycled. The recovered lignin also serves as fuel in our chemical recovery furnace, and greatly reduces our demand for non-renewable energy sources. The tan-colored pulp is then bleached to remove residual lignin and to whiten the fibers to our customers' specifications.Elemental chlorine was the dominant bleaching chemical in the cellulose fiber industry until the late 1980s, when the industry starting replacing it with chlorine dioxide. Chlorine dioxide is a very effective and environmentally favorable bleaching agent, but it's also unstable, and must be stored as a weak aqueous solution. This makes it necessary to manufacture and store chlorine dioxide solution at the mill site. The dominant chlorine dioxide generation processes reduce sodium chlorate to chlorine dioxide in a strong acid solution with methanol as a reducing agent. The desired reaction is:
3 NaClO3 + 2 H2SO4 + 0.85 CH3OH g 3 ClO2 + Na3H(SO4)2 + 2 H2O + 0.06 CH3OH + 0.52 HCOOH + 0.27 CO2
However, there are several side reactions, one of which can be summarized as:
3 NaClO3 + 2 H2SO4 + 1.5 CH3OH g 1.5 ClO2 + 0.75 Cl2 + Na3H(SO4)2 + 4.5 H2O + 1.5 CO2
Notice that in the desired reaction, every atom of chlorine is used to produce one molecule of chlorine dioxide. However, only half of the chlorine atoms in the side reaction produce chlorine dioxide with the remainder being consumed to produce elemental chlorine gas.
The ratio of chlorine dioxide molecules produced per molecule of sodium chlorate consumed is known as the conversion efficiency. Conversion efficiency is maximized by restricting generator acid and chlorate concentrations to a very narrow range. This task is made difficult by the lack of an on-line liquor strength sensor or analyzer. The only tools plant operators have to control reaction conditions are lab tests, look-up tables and their own experience and insight. In addition to controlling chemical concentrations, the operators also have to manage the concentration of suspended salt crystals, generator level, temperature and production rate. They must remain vigilant and keep an eye on secondary variables, such as generator vacuum and the ratio of steam to production rate. If any of these factors are allowed to drift too far from their targets, the chlorine dioxide can explosively decompose into chlorine gas and oxygen in an event euphemistically referred to as a "puff.” These puffs lift a heavy pressure relief plate on top of the generator and produce an intimidating boom when the plate slams back down. The equipment is designed to handle these events, and they aren't an exceptional hazard to people or equipment, but they do shut down production briefly, and the vibration has been known to damage pipes. These decomposition events can be caused by many factors, including high temperature, foam, contaminants in the water supply or chemical feeds, and even ultraviolet light.
This entire, complex interactive process is typically controlled manually by one operator. Our economic driver on this project was an increase in conversion efficiency, but we were well aware of the fact that this process is touchy and unforgiving and not popular among the operators. A significant secondary benefit of automation was to provide our coworkers with the tools needed to make this a less complex and more manageable process. Our hope was that this job will become more attractive and transferable to new recruits as our experienced operators begin to retire.
We disclosed to the operating team at the onset that this would be a learning process with the occasional setback. We planned to build the strategy in phases, and we were committed to keeping the strategy as simple as possible, but as complex as necessary to get the job done. And we did not restrict ourselves to process control subjects; the team also worked on other aspects of the operation, such as modifying the equipment and procedures related to defoamer addition, sampling and testing.
We began by automating level control with a simple PID controller that manipulates steam flow to the reboiler. This was the backbone of all the controls, since level variation affects every aspect of the generator operation —temperature, chemical concentrations and foaming. Next, we implemented ratio controllers to tie the chlorate and acid inlet flows to methanol flow. Methanol drives the reaction and is viewed as the "gas pedal” for this process. Ratio controllers eliminated the need for look-up tables to manually manage chemical feed rates during the frequent production rate changes, and they reduced the number of manipulated variables from three to one. We were struck by how immediate and predicable the production rate response was to methanol flow changes, and decided to build a production rate controller. These production rate and ratio controllers have completely replaced the look-up tables previously used by the operators to set up the generator for a desired production rate.
It became obvious to us while working on these initial controllers that the dominant process disturbance was the abrupt production rate changes made by the operators in an effort to manage the levels of their two large chlorine dioxide solution storage tanks. With all their other duties, it was not reasonable for us to expect them to make frequent small production rate changes throughout the day to avoid large changes later. The storage tanks are quite large and are intended to be a buffer between the bleach plant and the chlorine dioxide generator. We wanted a strategy that would allow the levels to fluctuate, and gradually adjust production rate to keep them within allowable limits.
The strategy we chose makes use of the small model predictive control (MPC) controller that comes included with our plant's new distributed control system. The MPC monitors both the rate of change in inventory and the actual levels. It has a zero rate-of-change target that is tuned quite slowly and level limit constraints that are tuned more aggressively. Along the way, we also implemented a strategy that automatically balances the two chlorine dioxide storage tanks. In this way, production rate changes are minimized as the storage level is allowed to meander slowly between high and low limits.
This controller has had a huge impact on the overall stability of the process — once our operators became comfortable with the increased ClO2 tank level movement. As one generator operator puts it, "The rate controller now ramps methanol flow to keep up with the bleach plant and keeps the rest of the generator stable.” The resulting improvement in temperature stability can be seen in Figure 1.
By this point we had captured much of the low-hanging fruit. The operators no longer had to manage the generator level or independently adjust feed flows, and the automated production and inventory control gave them freedom to work in the field for extended periods without worrying that their tanks were running empty or overflowing. As welcome as these controllers were, they did not produce the efficiency gains we were hoping for. It was still challenging to keep the generator liquor at the chlorate and acid concentrations needed to provide high conversion efficiency.
Figure 3. Simulations indicate that using the regression model should effectively avoid uncontrolled temperature drops that hurt efficiency and uncontrolled temperature climbs that often result in a decomposition event.
We decided to create an online estimate of generator liquor composition that could be used for feedback control. We drew upon earlier work published by Bruce Alison (Allison, B.J. "A Liquor Composition Control Strategy for Chlorine Dioxide Generators.” Proc., PacWest Conference, Jasper, AB, May 2004) with his equations reorganized to simplify computation. Our only project cost, other than our time, was a mass flowmeter purchased to measure incoming chlorate flow. The result was a state estimator that predicts generator concentrations using a chlorine mass balance and assumed conversion efficiency. Lab results are automatically time-shifted to match stored predictions and are used to correct the concentration and efficiency estimates.Two PID controllers were built to use these online concentration estimates as virtual sensors. "The estimators give us the ability to track how efficiently we're producing, which is a tool we haven't had before,” says another generator operator. "Now when we need to make any adjustments, we can see the impact on efficiency right away instead of waiting on a month-end accounting number.”
Early versions of the controllers worked well when the generator was stable, but were found to go into a "death spiral” under certain upset conditions. Our calculation was only capable of updating the assumed conversion efficiency every few hours when new lab results became available. However, we could clearly see that a drop in generator liquor temperature of more than a couple degrees was accompanied by a real and significant drop in conversion efficiency. During these periods, the controller would overestimate conversion efficiency and under-dose chemical. Chlorate and acid are known to influence the saturation temperature of the generator liquor and, therefore, a drop in their concentration causes a further loss of temperature and further reduction in efficiency. Our operators soon learned to avoid this death spiral by taking over and manually boosting the chlorate and acid flows to get the temperature back up.
We've recently augmented the estimator to predict temperature based on the current concentration estimates. We developed a regression model to relate our concentration estimates to generator temperature, then used the deviation between predicted and actual to adjust the conversion efficiency estimates (Figure 2). This provides a continuous feedback signal with which the estimator can adjust concentration and efficiency estimates in response to unexpected temperature fluctuations. Based on our simulations (Figure 3), this new feature should effectively avoid uncontrolled temperature drops that hurt our efficiency and uncontrolled temperature climbs that often result in a decomposition event.
We did not have a complete view of the final strategy when we began this project two years ago. The effort was trial and error, and included elements that we had to abandon along the way as our understanding of the problem improved. We could not have done this without the active involvement of the operators. All participated and helped improve the strategy in big and small ways, and a few key individuals jumped in with both feet to help with our experiments during their shift. We're currently working with other company sites to see if their chlorine dioxide generators could benefit from this strategy. And if we now appear to know what we're doing, it's due in no small part to the exceptional team of operators and production staff in Oglethorpe, Georgia.
Christopher McNabb is senior engineering advisor and Libby Berter is senior process engineer at Weyerhaeuser Co., Oglethorpe, Georgia.