Nuclear Security, Part IIFission Basics
By Béla Lipták, PE, control consultant
In the November column, (Nuclear Plant Security and Cyber Terrorism), I wrote about cyber terrorism and how process control can protect nuclear power plants or the chemical industry against it. Now I will discuss the overall topic of the nuclear power plant operation and the use of process control to protect against nuclear accidents. I will first describe the process and the reasons why some of the 100 accidents occurred in the past.
But first we need to familiarize ourselves with the basics of the nuclear power generation process, because we can only control a process if we fully understand it!
History
In 1905, Albert Einstein identified the relationship between matter and energy as e = mc2. In 1923, he received the Nobel Prize in physics for it. It took 103 years to prove his theory, but this year, he was finally proven right.1
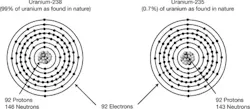
Figure 1. The atomic structure of a stable (238U) and an unstable (235U) isotope of uranaium. Only unstable isotopes can be used as nuclear fuel.
The concept of a nuclear reactor utilizing the chain reaction of fission was developed by the Hungarian-American2 scientist Leó Szilárd in 1933. He patented the process and later (1942) demonstrated it while working with a team of scientists headed by the Italian-American Enrico Fermi. This chain reaction was later used by the team of the German-American L. Robert Oppenheimer during the Manhattan Project in building the first atomic bombs.
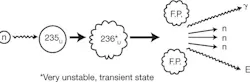
Figure 2: In a nuclear reactor, a slow thermal neutron enters the fissionable fuel (235U) and splits that atom while energy (E) and 2 or 3 neutrons are released. Some of these neutrons are “thermal” and others are “fast” having different energy levels.
Another way to release the energy of the atom was taken by the Hungarian-American scientist, the Hungarian-American Edward Teller in 1952, who proved that the fusion of hydrogen atoms can also be used to initiate a chain reaction. Teller also participated in the Manhattan Project and wanted to develop this weapon only to be used as a deterrent and later did all he could to prevent its use on civilian targets. In the last years of his life, he became convinced that nuclear weapons threaten human civilization and became one of the advocates of total nuclear disarmament. Teller also understood the great importance of process control and wrote the preface to the first edition of my Instrument Engineers’ Handbook3.
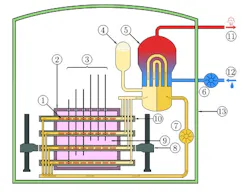
Figure 3: Schematic Diagram of a CANDU reactor:
The primary loop is in yellow and orange, the secondary in blue and red. The cool heavy water in the calandria can be seen in pink, along with partially-inserted shutoff rods.
(Courtesy of Wikipedia http://en.wikipedia.org/wiki/CANDU
Here, as an energy source, I will not discuss this fusion process, (which occurs in the sun), because that process operates at millions of degrees temperature and, therefore, is unpractical on this planet. I will just mention that to date, controlled fusion has only been achieved in experimental devices although a large fusion reactor is under construction in France with international support.
Understanding Nuclear Fission
An atom is composed of a central nucleus consisting of protons, neutrons and other particles plus of electrons orbiting in shells around the nucleus at discrete energy levels. These are referred to as electron shells. The proton has a positive charge equal to that of the electron and a mass, which is a couple of thousand times greater. The neutron has no electric charge and has a mass similar to that of the proton. When basic particles combine to form an atom, a certain amount of mass is converted into the binding energy of the atom, which is needed to hold the nucleus together. As I noted earlier, Einstein defined this conversion by the equation e = mc2, where e is the energy, m is the mass, and c is the velocity of light in a vacuum.
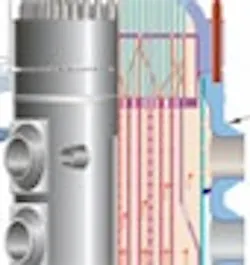
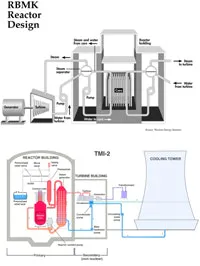
Figure 4: A schematic (top) and a more detailed view (bottom) of the reactor design which in 1986 at Chernobil caused the most severe nuclear accident to date. The design is referred to as the RBMK (Reactor Bolohoj Moshosztyl Kanalnyj) reactor.
In chemical reactions, the changes occur in the electron shells. In nuclear fusion, the release of energy is caused by the change in the nuclei as atomic particles are fused together. In fission, mass is converted into energy and energy is released, because the atoms of isotopes are split. Fission events release more than 2 million times more energy per event than do chemical reactions.
Atoms are neutral when their number of electrons equals the number of protons within their nucleus. When the number of electrons differs from the number of the protons, they have an electric charge and are called ions which can “chemically” combine with other ions of opposite charge.
Different elements have different numbers of protons (atomic numbers) in their nuclei and atoms of the same element can have different atomic masses because they may contain different numbers of neutrons. These are called isotopes (Figure 1). The stable isotopes have definite ratios of neutrons to protons in their nuclei (U-238), while unstable isotopes (U-235) do not.
During fission, a heavier unstable nucleus splits into two or more lighter nuclei, while releasing a substantial amount of energy. Fissionable materials include the naturally occurring isotope 235U4 and the man made isotope 239Pu5. Fission is initiated when a free neutron of the proper energy (thermal neutron) is captured by the nucleus of a fissionable atom. The most common way of generating thermal neutrons is to allow neutrons from a source—reactor, accelerator or spontaneous fission neutron emitter—to diffuse outward through a large block or tank of very weakly absorbing moderator. When the nucleus captures a thermal neutron, it will "split" producing two or more fission products (atoms of different elements formed from the protons, neutrons, and electrons originally comprising the original nucleus before its fission) plus two or three free neutrons and a tremendous amount of energy (Figure 2).
The emission of two or three free neutrons can split other unstable atoms (produce other fission events), which in turn will cause the emission of even more energy and more free neutrons. Within a few generations, the total amount of energy and the number free neutrons can become tremendous, sufficient to cause a nuclear explosion. For sustained chain reaction, it is necessary to moderate the process, to capture the “fast neutrons” while utilizing the energy released by the neutrons in the thermal range.
Reactor Designs
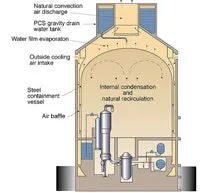
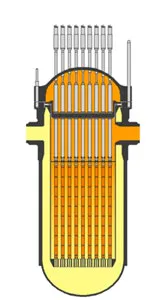
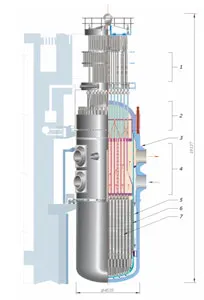
Today, there are basically three more advanced nuclear power plant designs in use. One is the CANDU system developed in Canada using heavy water moderator tubes (Figure 3). This design is similar to the Chernobyl RMBK design (Figure 4) only in that it uses pressure tubes instead of a pressure vessel, which facilitates on-line refueling, but otherwise it is much safer, because it contains much more cold heavy water. The General Electric design is a direct design (the moderator and the source of the steam to the turbine-generators is the same water). The third is the Westinghouse indirect design (Figure 5) (The high-pressure water in the reactor is the moderator and the coolant) in which the heat from the pressurized moderator water is used to boil the secondary water that is used to generate the steam for the turbines. This indirect Westinghouse AP1000 design is also the basis of the French EPR (Figure 6), and the Russian VVER1000 (Figure 7).
Figure 5: Diagram of AP600/AP1000 passive safety systems .
(Courtesy: Wikipedia)
Figure 6: The French EPR design (Courtesy of Wikipedia)
Figure 7: WWER-10ff (also VVER-1000 as a direct transliteration from Russian ВВЭР-1000). WWER-1000 (Water-Water Energetic Reactor, 1000 megawatt electric power) is a Russian nuclear power reactor of PWR type. (Courtesy of Wikipedia)
I will continue this series in the March issue.
Béla Lipták, PE, control consultant, is also editor of the Instrument Engineers’ Handbook and is seeking new co-authors for the for coming new edition.